 Getting cells to revert to a stem-like state – creating so-called induced pluripotent stem (iPS) cells – was a true revolution, but the technique invented in 2006 is only half the game. The first challenges include getting enough adult cells to undergo the “reprogramming” in culture to be of use and removing those traces of “priming” that distinguish them from true embryonic stem cells. The second is keeping them in the iPS state – that is, holding back their urge to differentiate – in lab conditions. And then there is the challenge of directing their differentiation in the way that you want, especially if you want to lure them down one of the very early developmental paths.
Getting cells to revert to a stem-like state – creating so-called induced pluripotent stem (iPS) cells – was a true revolution, but the technique invented in 2006 is only half the game. The first challenges include getting enough adult cells to undergo the “reprogramming” in culture to be of use and removing those traces of “priming” that distinguish them from true embryonic stem cells. The second is keeping them in the iPS state – that is, holding back their urge to differentiate – in lab conditions. And then there is the challenge of directing their differentiation in the way that you want, especially if you want to lure them down one of the very early developmental paths.
 For Dr. Jacob (Yaqub) Hanna, the way forward lies in taking a step back and asking the fundamental questions: How does reprogramming work on the basic molecular level? Why does the reprogramming process – inserting four genes into adult cells – create cells that are almost, but not quite, the equivalent of embryonic stem cells? Why is it that mouse embryonic stem cells tend to be stable and easily manipulated in the lab, but human stem cells stubbornly insist on differentiating or degrading, as well as resisting methods that have been shown to work on mouse cells?
For Dr. Jacob (Yaqub) Hanna, the way forward lies in taking a step back and asking the fundamental questions: How does reprogramming work on the basic molecular level? Why does the reprogramming process – inserting four genes into adult cells – create cells that are almost, but not quite, the equivalent of embryonic stem cells? Why is it that mouse embryonic stem cells tend to be stable and easily manipulated in the lab, but human stem cells stubbornly insist on differentiating or degrading, as well as resisting methods that have been shown to work on mouse cells?
 That last question has turned out to be crucial: It is by comparing mouse and human cells that Hanna and his lab group have gained an understanding of the genetic pathways in human cells that drive differentiation, and this understanding led them to create “better iPS cells.” We wrote about these “naïve” cells, which appear to be closer to the earliest embryonic stem cells, around a year ago saying: “The next step, of course, is directing the differentiation of the embryonic tissue to produce specific human tissues and organs. Hanna and his group are already on it.”
That last question has turned out to be crucial: It is by comparing mouse and human cells that Hanna and his lab group have gained an understanding of the genetic pathways in human cells that drive differentiation, and this understanding led them to create “better iPS cells.” We wrote about these “naïve” cells, which appear to be closer to the earliest embryonic stem cells, around a year ago saying: “The next step, of course, is directing the differentiation of the embryonic tissue to produce specific human tissues and organs. Hanna and his group are already on it.”
We are now pleased to report progress in that area. The cells that Hanna and his group have created in the lab in the course of their latest research are some of the most challenging: primordial germ cells, the precursor cells that appear just a week or two into embryonic development and eventually give rise to the sperm and ova. A group in Japan had produced them in mouse cells, but that feat was proving particularly tricky to reproduce in human cells.
 Using the naïve cell technique they invented, Hanna and his team went on to create human primordial germ cells – in quantity. “We can produce enough of these cells in the petri dish to use them for further research,” says Hanna. “One of the most important things we discovered is that the process is regulated by a different master gene in humans than in mice. This shows that we need to learn more about how these cells are produced in humans.”
Using the naïve cell technique they invented, Hanna and his team went on to create human primordial germ cells – in quantity. “We can produce enough of these cells in the petri dish to use them for further research,” says Hanna. “One of the most important things we discovered is that the process is regulated by a different master gene in humans than in mice. This shows that we need to learn more about how these cells are produced in humans.”
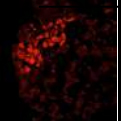
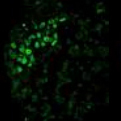
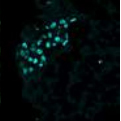
Images: Clusters of human embryonic stem cells that were differentiated to an early germ cell (PGC) state. Each color reveals the expression of a different gene. (top to bottom) NANOS3, NANOG, OCT4 and all three combined in a single image