"The more one does and sees and feels, the more one is able to do, and the more genuine may be one's appreciation of fundamental things like home, and love, and understanding companionship." -Amelia Earhart
It's been another fantastic week for us here at Starts With A Bang! This past week, we've had a blast covering a wide range of topics, including:
- The Amazing Spinning Electron (for Ask Ethan),
- The Top 5 Spectacles from Eurovision 2014 (for our Weekend Diversion),
- A Perfect Ten on the Celestial Equator, M10 (for Messier Monday),
- Why you're not alone, but probably wrong if you don't believe in cosmic inflation,
- What Scientific Arrogance Really Looks Like, and
- The Physics of Hot Pockets (for Throwback Thursday).
As always, I've hosted synopses of all these articles here on Scienceblogs for you to comment on and use as a forum, and you've responded with some truly great (and thought-provoking) ones! Without further ado, here are -- along with my responses -- your Comments of the Week!
Images credit: R. Nave of http://hyperphysics.phy-astr.gsu.edu/hbase/molecule/hmol.html.
From Peter Krahulik on the topic of how electrons allow hydrogen atoms to bond together (or not): "I like your spin explanation very much, but I want to ask the following: Would it be possible to collect a lot of hydrogen atoms exclusively with aligned spins (not bonding) to obtain atomic hydrogen gas? And what properties would it then have?"
It's very tricky when you take two isolated quantum systems, bring them together, and watch them interact. You make the assumption that because something is in a particular quantum state at some initial time, it isn't free to evolve as time moves forward. Under most conditions, when you place two free hydrogen atoms together, they're simply going to bind using the standard, bonding molecular orbital. Why? It's the most energetically favorable place to be, and -- typically -- there's nothing stopping them from going there. If you have two free hydrogen atoms, the electrons will tend to occupy the bonding orbitals, as they're lower in energy. But what if you're part of a more complicated molecule?
In this case -- which often happens in say, benzene rings -- there may be something causing a drop in electron density in the space between the two hydrogen atoms that are brought close together. In this case, because of other interactions, some pairs of hydrogen atoms may be forced into antibonding states, where the electrons cannot occupy the lower-energy bonding state, and instead occupy the higher-energy antibonding one. Fun fact: the energy difference between two free hydrogens and (greater energy) antibonding H2 is greater than the energy difference between two free hydrogens and (lower energy) bonding H2! Work on understanding bonding and antibonding molecular orbitals was such a powerful tool it was awarded the 1981 Nobel Prize in Chemistry.
 Image credit: Os of http://eurovision-addict.blogspot.com/2014/01/france-2014-song-titles.h….
Image credit: Os of http://eurovision-addict.blogspot.com/2014/01/france-2014-song-titles.h….
From Peter on the subject of this year's Eurovision song contest: "Glad you liked it, but why no comment on France, ‘Moustache’ has to be one of the craziest entries!"
You know, one of the pleasures of music, art, and spectacles in general is that one person's trash is another person's treasure. I didn't understand how Poland and Switzerland weren't in the top 10, I didn't understand how Armenia finished as high as 4th, and I thought it was catastrophic that Latvia didn't make the finale. But one of the things I did agree with was that there were 25 entries in the Eurovision finals that I found more enjoyable than France's. And one other thing I agreed with was David Hurn's comments about Conchita Wurst/Austria's victory:
"I am glad you enjoyed our spectacular Eurovision. Graham Norton is definitely the icing on the cake.
The best song may have not won, but the best sentiment did."
I'm smiling even now as I read that again.
From Sinisa Lazarek on the topic of cosmic inflation: "You wrote 2 articles about inflation with more or less same text and charts as this one, just 2 months ago. And they were the same as articles 2 months before that. The only news is BICEP2, but it’s not news anymore. So the question is why keep recycling?"
You know, this is something I struggle with. On the one hand, there are so many outstanding things to write about, and -- as you may have noticed -- I've been writing a lot more recently. For a long time, we were doing three original science posts a week here, along with one diversion on the weekend. More recently, we've been doing five original ones, a comments of the week on top of that and still the weekend diversion. And so when news breaks -- as it did this week about the BICEP2 team's results possibly being due to galactic foregrounds rather than cosmic inflation -- I'm more inclined that I was before to swoop in and say the important things that I think people ought to consider, especially if I don't see them said anyplace else.
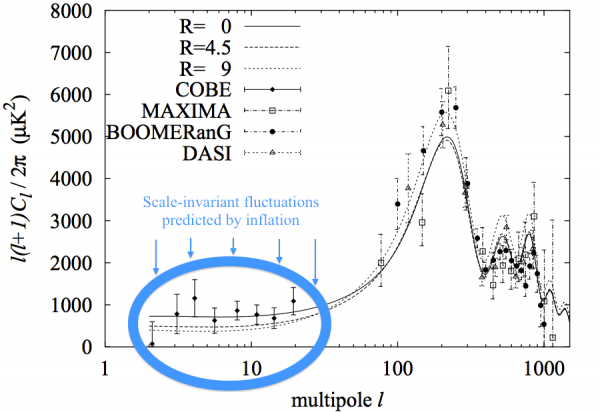 Image credit: Takeo Moroi & Tomo Takahashi, from http://arxiv.org/abs/hep-ph/0110096; annotations by me (in blue).
Image credit: Takeo Moroi & Tomo Takahashi, from http://arxiv.org/abs/hep-ph/0110096; annotations by me (in blue).
When it came to this particular topic -- and I'll confess to also being spurred on by a horrific John Horgan piece I read (and refuse to link to) -- I really wanted to address the full suite of evidence we had for cosmic inflation, and why even if the BICEP2 results turn out not to provide any evidence for inflation, it's still the leading theory of the origin of our observable, physical Universe for many good, solid, compelling reasons. I'm sorry if you didn't learn anything new from reading it, but I hope there are at least a few others out there who did. Although I always appreciate and welcome input from all, I try not to self-assess the quality of my posts from the happiness of my commenters!
 Image credit: Randall Munroe of xkcd, via http://xkcd.com/435/.
Image credit: Randall Munroe of xkcd, via http://xkcd.com/435/.
From Jason Taylor on the topic of scientific arrogance: "IMO physicists as a whole aren’t arrogant at all! It is non-scientists who are so arrogant that they don’t change their minds, don’t consider new data, don’t want to know the truth, all because they know “deep inside” that they are right."
First off, I do totally agree with the sentiment that if you cannot change your mind when presented with evidence that your previous conception or understanding of a phenomenon is incomplete, incorrect or invalid, what you are doing is the antithesis of good science. But let's not pretend that you need to be a scientist to do just that, and let's not pretend either that all scientists do, in fact, do that themselves. Fred Hoyle spent the last 40 years of his life adding more and more free parameters to his theory of the steady-state Universe to avoid accepting the Big Bang. Pavel Kroupa has repeatedly stated that even if they experimentally create dark matter in the laboratory he still won't accept that it exists as some 25-30% of the energy in the cosmos. And Halton Arp spent his entire life claiming that quasar redshifts were quantized and intrinsic properties of such objects, even though all evidence indicating such went away in the 1970s and never returned.
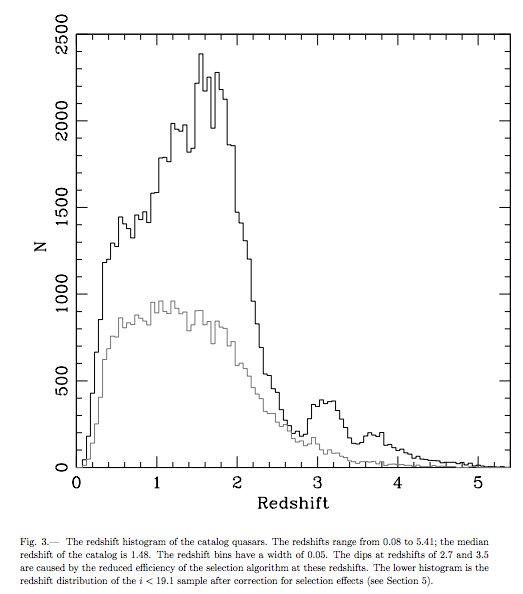 Image credit: Redshift (non-)quantization data, via Donald Schneider et al., 2007, via http://arxiv.org/abs/0704.0806.
Image credit: Redshift (non-)quantization data, via Donald Schneider et al., 2007, via http://arxiv.org/abs/0704.0806.
It isn't about whether you have a Ph.D., do research, teach, or otherwise practice science or not. It's about whether you can recognize the value of other people's work, knowledge, and expertise, and incorporate that into your own Universe. But I do agree with you about people who just know they are right about matters that are not verified in their favor. After all, I did write:
There’s a fundamental difference between scientific knowledge — knowledge that was obtained through experiment and observation of real, verifiable and reproducible phenomena — and what I’ll just call unverified or unverifiable claims. The former is the best source of knowledge that we have; the latter is ignorance falsely posing as knowledge, perhaps the greatest enemy of knowledge humanity has ever known.
 Image credit: Missouri Southern State University, via http://www.mssu.edu/academics/programs/philosophy.php.
Image credit: Missouri Southern State University, via http://www.mssu.edu/academics/programs/philosophy.php.
Also, on this same topic, I really enjoyed what Cleon Teunissen had to say, and wanted to share it with you, concerning Neil deGrasse Tyson and his comments on philosophy:
So what is deGrasse Tyson so dismissive about? What he rejects is ill-conceived questions. As I understand his point he rejects questions that are set up in such a way that effectively you’re casting yourself adrift.
Admittedly, in the history of philosophy some thinkers have done just that. They were just going nowhere.
By the looks of it deGrasse Tyson uses a sweeping redefinition of the word ‘philosophy’: to indulge in meaningless meanderings.
I disagree with that redefinition.
So do many philosophers of both the academic and armchair variety. In particular, probing the limits of knowledge and knowability is something that science cannot (yet) inform us about, and so if we're interested in thinking about that question at all, philosophy is an absolute must.
And finally...
 Image credit: The Pierateers of http://www.pierate.co.uk/2010/03/its-high-time-for-pie-time.html.
Image credit: The Pierateers of http://www.pierate.co.uk/2010/03/its-high-time-for-pie-time.html.
From dot tilde dot on the physics of hot pockets: "i put a pan of water into the oven to have steam when baking bread. i guess it helps to transport the energy into the cold loaf. is that a correct explanation for why my bread bakes faster and gets a slightly softer crumb with the steam?"
So I'm going to let you in on a little secret: I really enjoy cooking in general, but baking is by far the most common task that I'm simply no good at. Pie crusts, cakes, breads, you name it; if you need to bake it, you probably shouldn't ask me. (The one thing I can do is make a souffle rise!) That said, as best as I understand it, baking bread leavens because -- as the temperature rises -- the yeast cells consume the sugars in the bread at a faster and faster pace, until it gets too hot and they die. When yeast cells metabolize those sugars, they produce water and carbon dioxide, and the water converts into steam in the hot oven. The steam and carbon dioxide react together to inflate the bread, and if you add extra steam to the mix (for the first few minutes your bread is in the oven), you can add some extra "spring" to your bread.
 Image credit: The Anvil Proton steam injected convection oven, via One Fat Frog restaurant equipment.
Image credit: The Anvil Proton steam injected convection oven, via One Fat Frog restaurant equipment.
After all, steam-injection ovens are a thing for a reason! But it's not as much about transporting energy -- I don't think -- as it is for adding extra steam that can help the loaf leaven. But if there's one thing this week that I'm wrong about, it might be that. Any baker-scientists in the house?
Thanks for a great slew of comments this week (there are many more good ones on this post in particular), and I'll see you back here next week for more science, more comments and more Starts With A Bang!


I presume this is the John Horgan article on inflation that Ethan refused to link to. I also note that Ethan has not mentioned Resonaances, where Jester has blogged critically about BICEP2.
When it comes to scientific arrogance, I think it's wrong to censor opposing views and contrary evidence.
I'm all for open discussion of even things in which one does not see merit, but when discussion for discussion's sake occurs, as it can and does occur when one gets into the `if'' stages, well, I can see where scientists would finally just `throw up their hands' and move on to other things.
It's not censorship to refuse to link to an article that's in plenty of other places on the web, though. Or if it is, every single one of you is censoring my comments on how not to present polyamory to your children.
It's not as though google is a top-secret thing, so that John Duffield is among the few people who can find an article given the name of the author and the subject. But linking tells google "this is a good or interesting article," and there are sound reasons not to endorse things we strongly disagree with.
Není mi to moc jasné, ale zvětšením počtu elektronů v blízkosti atomu vodíku dojde při pozorování tohoto atomu k jejich ( elektronů ) uspořádání do určitých normálně neexistujících orbitalů?? Elektrony si vytvoří vlastní orbitaly??
aha, takže jde vlastně o vytvoření orbitalů z elektronů bez přítomnosti kladného náboje, tj. jak se elektrony navzájem uspořádají do určitých slupek...
stane se něco podobného u většího množství protonů kolem jádra vodíku??Jak se uspořádají protony??
Je možné totéž změřit u složitějších atomů než je vodík a zároveň u molekul??